|
Miltenyi Biotec
anti integrin β5 Anti Integrin β5, supplied by Miltenyi Biotec, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/anti integrin β5/product/Miltenyi Biotec Average 90 stars, based on 1 article reviews
anti integrin β5 - by Bioz Stars,
2026-03
90/100 stars
|
Buy from Supplier |
|
Becton Dickinson
alexa fluor ® 647-conjugated anti-β5 integrin ab  Alexa Fluor ® 647 Conjugated Anti β5 Integrin Ab, supplied by Becton Dickinson, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/alexa fluor ® 647-conjugated anti-β5 integrin ab/product/Becton Dickinson Average 90 stars, based on 1 article reviews
alexa fluor ® 647-conjugated anti-β5 integrin ab - by Bioz Stars,
2026-03
90/100 stars
|
Buy from Supplier |
|
Abnova
rabbit anti-integrin β5  Rabbit Anti Integrin β5, supplied by Abnova, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/rabbit anti-integrin β5/product/Abnova Average 90 stars, based on 1 article reviews
rabbit anti-integrin β5 - by Bioz Stars,
2026-03
90/100 stars
|
Buy from Supplier |
|
Enzo Biochem
20s subunit α4  20s Subunit α4, supplied by Enzo Biochem, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/20s subunit α4/product/Enzo Biochem Average 90 stars, based on 1 article reviews
20s subunit α4 - by Bioz Stars,
2026-03
90/100 stars
|
Buy from Supplier |
|
Abnova
mouse anti-integrin β5  Mouse Anti Integrin β5, supplied by Abnova, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/mouse anti-integrin β5/product/Abnova Average 90 stars, based on 1 article reviews
mouse anti-integrin β5 - by Bioz Stars,
2026-03
90/100 stars
|
Buy from Supplier |
|
MyBiosource Biotechnology
anti- integrin β5 antibody  Anti Integrin β5 Antibody, supplied by MyBiosource Biotechnology, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/anti- integrin β5 antibody/product/MyBiosource Biotechnology Average 90 stars, based on 1 article reviews
anti- integrin β5 antibody - by Bioz Stars,
2026-03
90/100 stars
|
Buy from Supplier |
|
Biomol GmbH
polyclonal “anti-core” (subunits α5/α7, β1, β5, β5i, β7) (20s proteasome  Polyclonal “Anti Core” (Subunits α5/α7, β1, β5, β5i, β7) (20s Proteasome, supplied by Biomol GmbH, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/polyclonal “anti-core” (subunits α5/α7, β1, β5, β5i, β7) (20s proteasome/product/Biomol GmbH Average 90 stars, based on 1 article reviews
polyclonal “anti-core” (subunits α5/α7, β1, β5, β5i, β7) (20s proteasome - by Bioz Stars,
2026-03
90/100 stars
|
Buy from Supplier |
|
Onyx Pharmaceuticals
anti-β5 polyclonal antibody  Anti β5 Polyclonal Antibody, supplied by Onyx Pharmaceuticals, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/anti-β5 polyclonal antibody/product/Onyx Pharmaceuticals Average 90 stars, based on 1 article reviews
anti-β5 polyclonal antibody - by Bioz Stars,
2026-03
90/100 stars
|
Buy from Supplier |
|
Biomol GmbH
antiproteasome anti-β5  Antiproteasome Anti β5, supplied by Biomol GmbH, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/antiproteasome anti-β5/product/Biomol GmbH Average 90 stars, based on 1 article reviews
antiproteasome anti-β5 - by Bioz Stars,
2026-03
90/100 stars
|
Buy from Supplier |
|
Biomol GmbH
anti–x(β5) rabbit pab  Anti–X(β5) Rabbit Pab, supplied by Biomol GmbH, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/anti–x(β5) rabbit pab/product/Biomol GmbH Average 90 stars, based on 1 article reviews
anti–x(β5) rabbit pab - by Bioz Stars,
2026-03
90/100 stars
|
Buy from Supplier |
|
Abnova
pab anti-β5 (h00003693d01p [rabbit  Pab Anti β5 (H00003693d01p [Rabbit, supplied by Abnova, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/pab anti-β5 (h00003693d01p [rabbit/product/Abnova Average 90 stars, based on 1 article reviews
pab anti-β5 (h00003693d01p [rabbit - by Bioz Stars,
2026-03
90/100 stars
|
Buy from Supplier |
|
Biomol GmbH
anti-proteasome subunit β5 β2  Anti Proteasome Subunit β5 β2, supplied by Biomol GmbH, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/anti-proteasome subunit β5 β2/product/Biomol GmbH Average 90 stars, based on 1 article reviews
anti-proteasome subunit β5 β2 - by Bioz Stars,
2026-03
90/100 stars
|
Buy from Supplier |
Image Search Results

Journal: bioRxiv
Article Title: Tumor-resident regulatory T cells in pancreatic cancer express the αvβ5 integrin as a targetable activation marker
doi: 10.1101/2023.05.24.542137
Figure Lengend Snippet: a Representative confocal images of αvβ5 integrin + CD4 + Foxp3 + T cells (left) and NRP-1 + CD4 + Foxp3 + T cells (right) in the PDAC tissue of KPC-derived orthotopic PDAC mice. Arrows indicate αvβ5 integrin + or NRP-1 + Tregs (magenta). Green, CD4; red, Foxp3; blue, DAPI. The boxed areas are magnified. Scale bars, 20 μm. b A representative flow cytometry analysis showing the proportion of CD8 + T cells, CD4 + CD25 neg T cells (non-Tregs), CD4 + CD25 + Tregs that are positive for αvβ5 integrin, NRP-1, or both in KPC-derived orthotopic PDAC tumors. c Bar diagrams that summarize the findings from ( b ) and Supplementary Fig. 10. n = 5 per group. d Representative confocal images of αvβ5 integrin + Foxp3 + CD4 + Tregs and NRP-1 + CD4 + Tregs in human PDAC and spleen. Magenta, αvβ5 integrin or NRP-1; green, CD4; red, Foxp3; blue, DAPI. Scale bars, 20 μm. e The number of αvβ5 integrin-positive and NRP-1-positive CD4 + Foxp3 + Tregs was counted under a confocal microscope and the % positivity was calculated. n = 3. Statistical analysis, one-way ANOVA ( c ) and Welch’s test ( e ); p = 0.0003 ( c, left, CD8 vs Tregs), p = 0.0169 ( c, left, Non-Tregs vs Tregs), p = 0.0002 ( c, left, Tregs vs Spleen Tregs), p = 0.0003 ( c, center, CD8 vs Tregs), p = 0.0123 ( c, center, Non-Tregs vs Tregs), p < 0.0001 ( c, center, Tregs vs Spleen Tregs), p = 0.0008 ( c, right, CD8 vs Tregs), p = 0.0109 ( c, right, Non-Tregs vs Tregs), p = 0.0019 ( c, right, Tregs vs Spleen Tregs), p = 0.0071 ( e, left), p = 0.4858 ( e, right). Error bars, mean ± standard error. * p < 0.05; ** p < 0.01; *** p < 0.001; **** p < 0.0001; N.S., not significant.
Article Snippet: After removing dead cells with a Dead Cell Removal kit (Miltenyi Biotec), the cells were treated with a PE-conjugated anti-CD25 Ab (Miltenyi Biotec), PE-conjugated anti-CCR8 Ab (eBioscience), and/or Alexa Fluor ® 647-conjugated
Techniques: Derivative Assay, Flow Cytometry, Microscopy

Journal: bioRxiv
Article Title: Tumor-resident regulatory T cells in pancreatic cancer express the αvβ5 integrin as a targetable activation marker
doi: 10.1101/2023.05.24.542137
Figure Lengend Snippet: a-e CD4 + T cells isolated from the spleens of healthy C57B6129SF1/J hybrid mice were expanded for 3 days in vitro in the presence of KPC-derived PDAC cells. Flow cytometry was performed for subsequent analyses. ( a ) Expression of αvβ5 integrin and NRP-1 on CD4 + CD25 + Tregs expanded with or without the PDAC cells. n = 4 per group. ( b ) Expression of αvβ5 integrin and NRP-1 on CD4 + CD25 + Tregs and non-Treg CD4 + CD25 neg T cells expanded with the PDAC cells. n = 3 per group. ( c ) FAM-iRGD binding to the non-Tregs (blue line) and Tregs (red line) shown in ( b ). The bar diagram summarizes the median fluorescence intensity (MFI) from 4 independent experiments. ( d ) Dose-dependent inhibition of FAM-iRGD binding by an anti-αvβ5 integrin blocking Ab to Tregs that were expanded in the presence of PDAC cells. Values were normalized against isotype control. n = 3 per group. Statistical analysis was performed between the isotype control and anti-αvβ5 integrin values. ( e ) Tregs and non-Tregs were expanded in the presence of PDAC cells with or without iRGD. Apoptosis was quantified by measuring annexin V and 7-AAD double positive cells by flow cytometry. n = 3. f, g In vitro binding of FAM-iRGD to non-Treg CD4 + CD25 neg T cells (blue line) and CD4 + CD25 + Tregs (red line) isolated from the PDAC tissue ( f ) or the spleen ( g ) of KPC-derived orthotopic PDAC mice. The bar diagrams summarize the MFI from 4 independent experiments. h Representative confocal images of Foxp3 + T cells (red) in the PDAC tissue of KPC-derived orthotopic PDAC mice that received an intravenous injection of FAM-iRGD (green). Blue, DAPI. Arrows indicate Tregs positive for iRGD. The boxed area is magnified. Scale bars, 20 μm. Statistical analyses; Mann-Whitney U test ( a , c , f , g ), Welch’s t test ( b , e ), and one sample Wilcoxon signed rank test ( d ); p = 0.0286 ( a , top), p = 0.3429 ( a , bottom), p = 0.0058 ( b , top), p = 0.0086 ( b , bottom), p = 0.0017 ( c ), p = 0.7272 ( d , 0.3), p = 0.1766 ( d , 1.0), p = 0.0052 ( d , 3.0), p = 0.0087 ( e , top), p = 0.6583 ( e , bottom), p = 0.0286 ( f ), p = 0.6857 ( g ). Error bars, mean ± standard error; * p < 0.05; ** p < 0.01; N.S., not significant.
Article Snippet: After removing dead cells with a Dead Cell Removal kit (Miltenyi Biotec), the cells were treated with a PE-conjugated anti-CD25 Ab (Miltenyi Biotec), PE-conjugated anti-CCR8 Ab (eBioscience), and/or Alexa Fluor ® 647-conjugated
Techniques: Isolation, In Vitro, Derivative Assay, Flow Cytometry, Expressing, Binding Assay, Fluorescence, Inhibition, Blocking Assay, Injection, MANN-WHITNEY
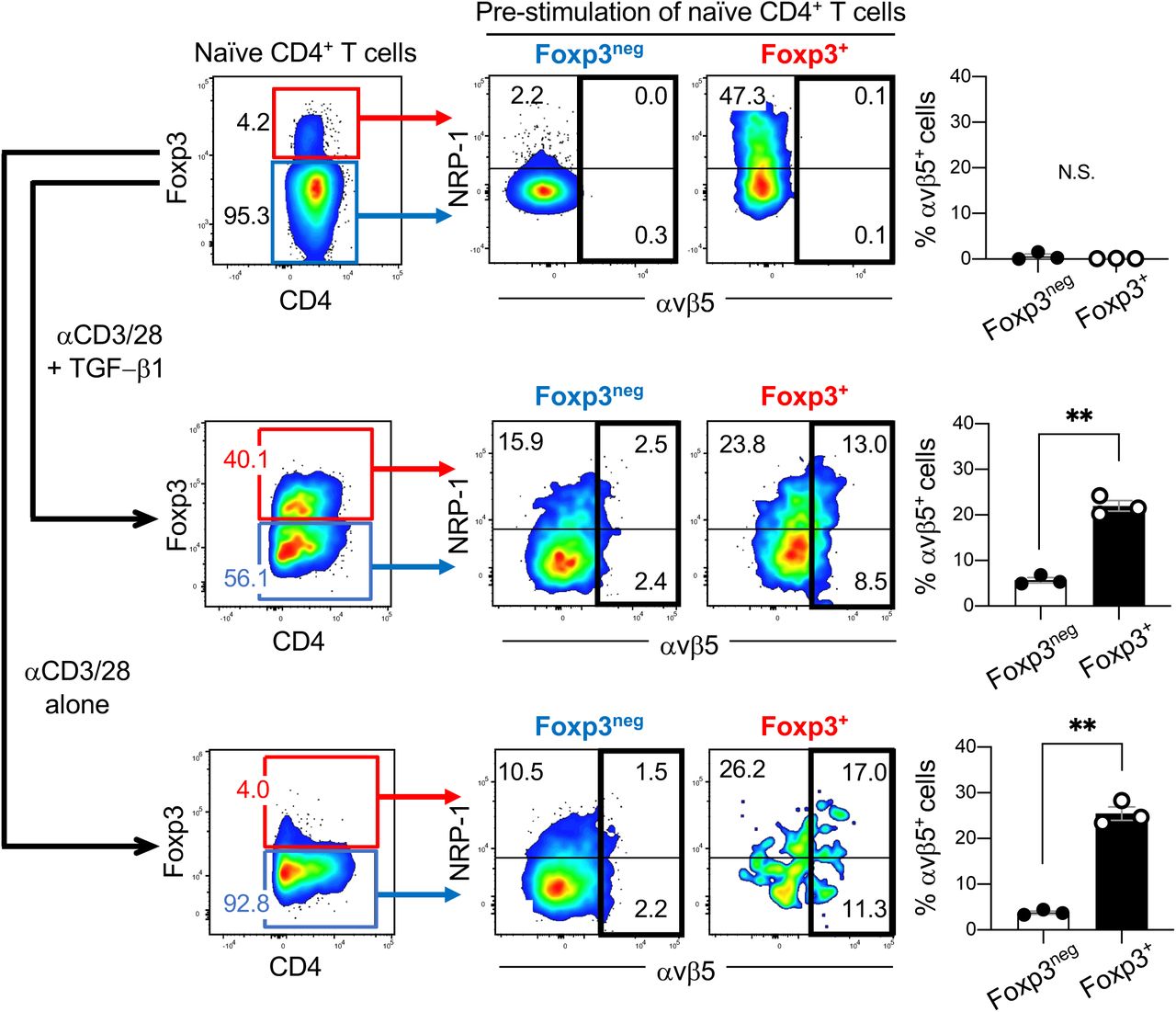
Journal: bioRxiv
Article Title: Tumor-resident regulatory T cells in pancreatic cancer express the αvβ5 integrin as a targetable activation marker
doi: 10.1101/2023.05.24.542137
Figure Lengend Snippet: Naïve CD4 + T cells were isolated from the spleens of healthy C57B6129SF1/J hybrid mice by magnetically removing CD4 neg T cells and CD25 + T cells. The pool enriched for naïve CD4 + T cells was cultured in vitro with anti-CD3/CD28 Abs in the presence or absence of TGF-β1 for 3 days, and analyzed for αvβ5 integrin and NRP-1 expression by flow cytometry. ( Top row) Naïve CD4 + T cells (blue box) enriched from mouse splenocytes. A minor population of CD4 + Foxp3 + T cells was present (red box). ( Middle row ) Treating the pool in the top row with anti-CD3/CD28 Abs and TGF-β1 yielded approximately 40% of CD4 + Foxp3 + T cells (red box) and 56% of CD4 + Foxp3 neg T cells (blue box). ( Bottom row ) Treating the pool in the top row with anti-CD3/CD28 Abs alone did not change the proportion of the CD4 + T cells. Nearly 95% of the cells remained negative for Foxp3 (blue box). Representative dot plots showing the proportion of CD4 + Foxp3 + T cells (left panels) and the expression of αvβ5 integrin and NRP-1 on the indicated population are presented. The bar diagrams summarize the proportion of αvβ5 + cells in the indicated population. n = 3 per study. Statistical analysis, Welch’s t test; p = 0.4115 (top), p = 0.0013 (middle), p = 0.0032 (bottom). Error bars, mean ± standard error; ** p < 0.01; N.S., not significant.
Article Snippet: After removing dead cells with a Dead Cell Removal kit (Miltenyi Biotec), the cells were treated with a PE-conjugated anti-CD25 Ab (Miltenyi Biotec), PE-conjugated anti-CCR8 Ab (eBioscience), and/or Alexa Fluor ® 647-conjugated
Techniques: Isolation, Cell Culture, In Vitro, Expressing, Flow Cytometry

Journal: bioRxiv
Article Title: Tumor-resident regulatory T cells in pancreatic cancer express the αvβ5 integrin as a targetable activation marker
doi: 10.1101/2023.05.24.542137
Figure Lengend Snippet: a, b Naïve CD4 + T cells isolated from healthy mouse spleens were expanded in the presence of anti-CD3/CD28 Abs and TGF-β1 ( a ) or anti-CD3/CD28 Abs alone ( b ) for 3 days. The resulting populations were gated based on Foxp3 and CD25 expression (left panels). αvβ5 integrin and NRP-1 expression on Foxp3 + cells ( a , top row) and Foxp3 neg cells ( a , bottom row; b ) was analyzed by flow cytometry. The red and blue boxes gate CD25 + and CD25 neg cells, respectively. The bar diagrams summarize the proportion of αvβ5 integrin-positive cells in the indicated T cell population. n = 3. Statistical analysis, Welch’s t test; p = 0.0021 ( a , top), p = 0.0014 ( a , bottom), p < 0.0001 ( b ). c Flow cytometric analysis showing the proportion of αvβ5 integrin + cells among CD4 + CD25 + Foxp3 + iTregs induced by increasing concentrations of anti-CD3 Ab. n = 3. Statistical analysis, one-way ANOVA; p = 0.0088 (1 vs 3), p = 0.0088 (1 vs 10). d-f Naïve CD4 + T cells were stimulated with anti-CD3/CD28 Abs and TGF-β1 in the absence or presence of iRGD or iRGE. Flow cytometry was performed to quantify the proportion of CD4 + CD25 + Foxp3 + iTregs ( d ) and αvβ5 integrin + cells among the iTregs ( e ). Apoptosis of αvβ5 integrin + iTregs was quantified by measuring cleaved caspase 3 using flow cytometry ( f ). n = 3. Statistical analysis, one-way ANOVA ( d , e ) or Welch’s t test ( f ); p = 0.0042 ( d , None vs iRGD), p = 0.1774 ( d , None vs iRGE), p = 0.0027 ( e , None vs iRGD), p = 0.0529 ( e , None vs iRGE), p = 0.001 ( f ). Error bars, mean ± standard error; * p < 0.05; ** p < 0.01; **** p < 0.0001; N.S., not significant.
Article Snippet: After removing dead cells with a Dead Cell Removal kit (Miltenyi Biotec), the cells were treated with a PE-conjugated anti-CD25 Ab (Miltenyi Biotec), PE-conjugated anti-CCR8 Ab (eBioscience), and/or Alexa Fluor ® 647-conjugated
Techniques: Isolation, Expressing, Flow Cytometry

Journal: bioRxiv
Article Title: Tumor-resident regulatory T cells in pancreatic cancer express the αvβ5 integrin as a targetable activation marker
doi: 10.1101/2023.05.24.542137
Figure Lengend Snippet: a CD4 + CD25 + Foxp3 + T cells (nTregs) were enriched from the spleens of healthy C57B6129SF1/J hybrid mice by magnetically removing CD4 neg T cells and CD25 neg T cells. The middle two panels show the expression of αvβ5 integrin and NRP-1 on the nTregs (red box) and naïve CD4 + CD25 neg Foxp3 neg T cells (blue box) analyzed by flow cytometry. The bar diagram summarizes the proportion of αvβ5 integrin + cells among the two populations. n = 3. Statistical analysis, Welch’s t test; p = 0.4721. b The pool in ( a ) was treated with anti-CD3/CD28 Abs alone for 3 days (left panel). αvβ5 integrin and NRP-1 expression on Foxp3 + cells (top row) and Foxp3 neg cells (bottom row) was analyzed by flow cytometry. The red and blue boxes gate CD25 + and CD25 neg cells, respectively. The bar diagrams summarize the proportion of αvβ5 integrin + cells among the indicated T cell populations. n = 3. Statistical analysis, Welch’s t test; p = 0.0064 (top), p = 0.0035 (bottom). Error bars, mean ± standard error; ** p < 0.01; N.S., not significant.
Article Snippet: After removing dead cells with a Dead Cell Removal kit (Miltenyi Biotec), the cells were treated with a PE-conjugated anti-CD25 Ab (Miltenyi Biotec), PE-conjugated anti-CCR8 Ab (eBioscience), and/or Alexa Fluor ® 647-conjugated
Techniques: Expressing, Flow Cytometry

Journal: bioRxiv
Article Title: Tumor-resident regulatory T cells in pancreatic cancer express the αvβ5 integrin as a targetable activation marker
doi: 10.1101/2023.05.24.542137
Figure Lengend Snippet: Naïve CD4 + CD25 neg Foxp3 neg T cells were magnetically isolated from the spleens of healthy C57B6129SF1/J mice. The cells were stimulated with anti-CD3/CD28 Abs and TGF-β1 for 3 days to induce CD4 + CD25 + Foxp3 + iTregs. a-c Expression of CCR8 and αvβ5 integrin on the T cells before and after the stimulation. Representative dot plots from 3 or 4 separate studies are shown in ( a ). The bar diagram in ( b ) summarizes the proportion of CCR8 + cells (white bars) and αvβ5 integrin + cells (black bars) among each T cell population in ( a ). The Venn diagram in ( c ) summarizes the proportion of iTregs that expressed CCR8 and/or αvβ5 integrin. d-f Treg suppression assays were performed by co-culturing iTregs and Tconv (CD4 + and CD8 + ) at a 1 : 4 ratio in the presence of anti-CD3/CD28 Abs (TCR stimulation) for 3 days. We used iTregs that were enriched for CCR8 + iTregs ( d ) or CCR8 + iTregs that were either depleted or enriched for αvβ5 integrin + cells ( e ). The expression of CCR8 and αvβ5 integrin on the iTregs is shown in the representative dot plots. Proliferation of Tconv was analyzed by flow cytometry using Cell Trace Violet as shown in the representative histograms: Shaded, iTregs + Tconv (with TCR stimulation); black solid line, Tconv alone (with TCR stimulation); black dotted line, Tconv alone (no TCR stimulation). The bar diagrams in ( f ) summarize the values from ( d ) and ( e ) normalized to stimulated Tconv alone. n = 3. Statistical analysis, one-way ANOVA; p < 0.0001 ( b , CCR8 + , CD25 neg Foxp3 neg vs CD25 + Foxp3 neg and CD25 + Foxp3 neg vs CD25 + Foxp3 + ; αvβ5 + , CD25 + Foxp3 neg vs CD25 + Foxp3 + ), p = 0.0989 ( b , αvβ5 + , CD25 neg Foxp3 neg vs CD25 + Foxp3 neg ), p = 0.0003 ( f , CD4, CCR8 + vs αvβ5 neg CCR8 + ), p = 0.0002 ( f , CD4, CCR8 + vs αvβ5 + CCR8 + ), p < 0.0001 ( f , CD4, αvβ5 neg CCR8 + vs αvβ5 + CCR8 + ), p = 0.1048 ( f , CD8, CCR8 + vs αvβ5 neg CCR8 + ), p < 0.0001 ( f , CD8, CCR8 + vs αvβ5 + CCR8 + and αvβ5 neg CCR8 + vs αvβ5 + CCR8 + ). Error bars, mean ± standard error; *** p < 0.001; **** p < 0.0001; N.S., not significant.
Article Snippet: After removing dead cells with a Dead Cell Removal kit (Miltenyi Biotec), the cells were treated with a PE-conjugated anti-CD25 Ab (Miltenyi Biotec), PE-conjugated anti-CCR8 Ab (eBioscience), and/or Alexa Fluor ® 647-conjugated
Techniques: Isolation, Expressing, Flow Cytometry

Journal: The Journal of Biological Chemistry
Article Title: Inhibition of Proteasome Activity Induces Formation of Alternative Proteasome Complexes
doi: 10.1074/jbc.M116.717652
Figure Lengend Snippet: Proteasomal activators interact with 20S/26S proteasomes forming alternative proteasome complexes. A, gel filtration assay of PA28γ proteasome complexes in BZ-treated HeLa cells compared with control. Western blotting analysis of RPT6, β2, and PA28γ in the top panel shows representative images of the protein distribution in the different fractions separated by gel filtration. The bottom panel shows the quantification of the signal for PA28γ in BZ-treated cells and controls. For clarity, the intensity of the signal has been normalized to the maximum value of each curve, and the total areas under each curve were made equal. B, analysis of interaction of proteasomal activators with the 20S proteasome using co-immunoprecipitation (IP). Lysates of phLF were treated with BZ for 6 h or with solvent, lysed, and subjected to immunoprecipitation using an antibody against the 20S subunit α4. Co-immunoprecipitated proteins as well as total protein lysate (input, 10% of total volume) were separated by SDS-PAGE. Direct interaction of proteasomal activators PA28γ and PA200 with the 20S proteasome subunit α4 was visualized via immunoblotting. Representative results of experiments in cells from three different donors are shown.
Article Snippet: Membranes were incubated overnight at 4 °C with one of the following antibodies against PA28γ (sc-136025, 1:1000, Santa Cruz Biotechnology; BML-PW8190-0100, 1:2000, Enzo Life Sciences), PA200 (NBP2-22236, 1:3000, Novus Biologicals), PA28α (ab155091, 1:1000, Abcam),
Techniques: Filtration, Western Blot, Immunoprecipitation, SDS Page

Journal: PLoS Pathogens
Article Title: Serum bridging molecules drive candidal invasion of human but not mouse endothelial cells
doi: 10.1371/journal.ppat.1010681
Figure Lengend Snippet: (A-F) Effects of inhibiting αv integrin function with siRNA knockdown (A and B or)specific monoclonal antibodies (C-F) and on the endocytosis (A, C, D) and cell-association (B, E, F) of serum-coated C . glabrata . (G and H) Inhibition of gC1qR (with monoclonal antibody 74.5.2) and αv integrins has an additive effect on decreasing the endocytosis (G) but not cell-association of serum-coated C . glabrata (H). Results are the mean ± SD of 3 experiments, each performed in triplicate. Orgs/HPF, organisms per high power field; ns, not significant; * P < 0.05, ** P < 0.01, *** P < 0.001, **** P < 0.0001 by ANOVA with the Dunnett’s test for multiple comparisons (A, B, G, H) or the Student’s t-test (C-F).
Article Snippet: For the cells transduced with hC1QBP, 10 μg/ml of blasticidin (Gibco; # A1113903) was added to the medium 2 d post transduction to select for transduced cells and selection was maintained for 7 d. Expression of eGFP was determined by fluorescent microscopy and expression of gC1qR, integrin αv, and integrin β5 were verified via immunoblotting of whole cell lysates with an anti-gC1qR antibody (clone 60.11), anti- integrin αv antibody (MilliporeSigma; #AB1930), and
Techniques: Inhibition

Journal: PLoS Pathogens
Article Title: Serum bridging molecules drive candidal invasion of human but not mouse endothelial cells
doi: 10.1371/journal.ppat.1010681
Figure Lengend Snippet: (A and B) Endocytosis of C . glabrata coated with either human or mouse serum by the indicated endothelial cells after 45 min (A) and 180 min (B). (C) Endocytosis of C . glabrata coated with fresh human serum by mouse liver endothelial cells expressing human gC1qR, integrin αv, or integrin β5. Data are the mean ± SD of 3 experiments each performed in triplicate. HUVEC, human umbilical vein endothelial cell; orgs/HPF, organisms per high power field; ns, not significant; ** P < 0.01, **** P < 0.0001. *** P < 0.001, **** P < 0.0001 by ANOVA with the Dunnett’s test for multiple comparisons.
Article Snippet: For the cells transduced with hC1QBP, 10 μg/ml of blasticidin (Gibco; # A1113903) was added to the medium 2 d post transduction to select for transduced cells and selection was maintained for 7 d. Expression of eGFP was determined by fluorescent microscopy and expression of gC1qR, integrin αv, and integrin β5 were verified via immunoblotting of whole cell lysates with an anti-gC1qR antibody (clone 60.11), anti- integrin αv antibody (MilliporeSigma; #AB1930), and
Techniques: Expressing

Journal: Acta Naturae
Article Title: Changes in the Proteasome Pool during Malignant Transformation of Mouse Liver Cells
doi:
Figure Lengend Snippet: Chymotrypsin–like activity of proteasomes and the content of the proteasome subunits, nNOS , β–actine, and total protein in the clarified homogenates of mouse liver and induced liver tumors.
Article Snippet: The following reagents were used: Suc–LLVY–AMC and MG132 (Sigma, USA), anti–β–actin mouse mAb (Santa Cruz, Germany), anti–Rpt6 and anti–α1,2,3,5,6,7 mouse mAb , anti–X(β5), anti–LMP7(
Techniques: Activity Assay, Protein Concentration

Journal: Acta Naturae
Article Title: Changes in the Proteasome Pool during Malignant Transformation of Mouse Liver Cells
doi:
Figure Lengend Snippet: Western blot analysis of proteins in the clarified homogenates of the intact liver (1), liver with diffuse nodules (2), and hepatocellular carcinoma (3) using antibodies to proteasome subunits α1,2,3,5,6,7, Rpt6, LMP7(β5i), X(β5), and LMP2(β1i), nNOS and β-actin. Markers: carboanhydrase (29 kDa), ovalbumin (45 kDa), trypsin inhibitor (20 kDa), and β-galactosidase (116 kDa)
Article Snippet: The following reagents were used: Suc–LLVY–AMC and MG132 (Sigma, USA), anti–β–actin mouse mAb (Santa Cruz, Germany), anti–Rpt6 and anti–α1,2,3,5,6,7 mouse mAb , anti–X(β5), anti–LMP7(
Techniques: Western Blot

Journal: Journal of Virology
Article Title: Human Adenovirus Type 37 Uses α V β 1 and α 3 β 1 Integrins for Infection of Human Corneal Cells
doi: 10.1128/JVI.02019-16
Figure Lengend Snippet: Cross sections of human corneas labeled (in green) with antibodies against integrin α2 (P1E6), α3 (ASC-1), α4 (P4C2), α5 (P1D6), α6 (MP4F10), αV (272-17E6), β1 (P5D2), β3 (MHF4), β4 (422325), and β5 (H00051706-D01P), as well as secondary-antibody-only controls GAM Alexa 488 and GAR Alexa 488. Notice the absence of specific labeling of the cell surface with antibodies against integrin α4, α5, and β5. Bar, 25 μm. Nuclei are stained using DAPI (blue). Representative images from 3 cornea explants are shown.
Article Snippet: The integrin-specific antibodies used for flow cytometry, in situ immunohistochemistry, colocalization, binding, and infection competition analyses were the following: MAbs anti-α2 (clone P1E6 [mouse]), anti-α3 (clones P1B5 [mouse] and ASC-1 [mouse]), anti-α4 (clones P4C2 [mouse] and PS/2 [rat]), and anti-α5 (clone P1D6 [mouse]), all from Merck Millipore; anti-α6 MAbs (clones GoH3 [rat] and MP 4F10 [mouse]) from Abcam; MAb anti-αV (clone 272-17E6 [mouse]) from Thermo Fisher Scientific; MAb anti-β1 (clone P5D2 [mouse]) and PAb anti-β1 (AF1778 [goat]), both from R&D Systems; MAb anti-β3 (clone MHF4 [mouse]) from Abnova; MAb anti-β4 (clone 422325 [mouse]) from R&D Systems; and
Techniques: Labeling, Staining

Journal: Journal of Virology
Article Title: Human Adenovirus Type 37 Uses α V β 1 and α 3 β 1 Integrins for Infection of Human Corneal Cells
doi: 10.1128/JVI.02019-16
Figure Lengend Snippet: Flow cytometry analysis of integrin subunit expression on HCE cells. The following antibodies were used to detect respective integrin subunits: α2 (P1E6), α3 (P1B5), α4 (P4C2), α5 (P1D6), α6 (GoH3), αV (272-17E6), β1 (P5D2), β3 (MF4), β4 (422325), and β5 (H00003693-D01P). Red and green colors represent staining in the presence and absence of primary antibody, respectively. n = 2.
Article Snippet: The integrin-specific antibodies used for flow cytometry, in situ immunohistochemistry, colocalization, binding, and infection competition analyses were the following: MAbs anti-α2 (clone P1E6 [mouse]), anti-α3 (clones P1B5 [mouse] and ASC-1 [mouse]), anti-α4 (clones P4C2 [mouse] and PS/2 [rat]), and anti-α5 (clone P1D6 [mouse]), all from Merck Millipore; anti-α6 MAbs (clones GoH3 [rat] and MP 4F10 [mouse]) from Abcam; MAb anti-αV (clone 272-17E6 [mouse]) from Thermo Fisher Scientific; MAb anti-β1 (clone P5D2 [mouse]) and PAb anti-β1 (AF1778 [goat]), both from R&D Systems; MAb anti-β3 (clone MHF4 [mouse]) from Abnova; MAb anti-β4 (clone 422325 [mouse]) from R&D Systems; and
Techniques: Flow Cytometry, Expressing, Staining

Journal: Journal of Virology
Article Title: Human Adenovirus Type 37 Uses α V β 1 and α 3 β 1 Integrins for Infection of Human Corneal Cells
doi: 10.1128/JVI.02019-16
Figure Lengend Snippet: Effects of preincubating HCE cells with anti-integrin antibodies on HAdV-37 and HAdV-5 infection and binding. (A) Effect of antibodies against integrin α subunits on HAdV-37 and HAdV-5 infection. (B) Effect of antibodies against integrin β subunits on HAdV-37 and HAdV-5 infection. (C) Effect of antibodies against integrin α and β subunits on HAdV-37 and HAdV-5 binding. Infection was assayed by counting virus-positive cells by immunofluorescence, and binding was assayed by quantitating 35 S-labeled virus association with cells. The results are presented as percentages of the control (i.e., untreated cells). The following antibodies were used: α2 (P1E6), α3 (P1B5), α4 (P4C2), α5 (P1D6), α6 (GoH3), αV (272-17E6), β1 (P5D2), β3 (MHF4), β4 (422325), and β5 (H00003693-D01P). The anti-GD1a specific MAb EM9 was used as a positive control. Results are shown as a percentage of the value for the control. *, P < 0.05; **, P < 0.01; ***, P < 0.001. n = 3.
Article Snippet: The integrin-specific antibodies used for flow cytometry, in situ immunohistochemistry, colocalization, binding, and infection competition analyses were the following: MAbs anti-α2 (clone P1E6 [mouse]), anti-α3 (clones P1B5 [mouse] and ASC-1 [mouse]), anti-α4 (clones P4C2 [mouse] and PS/2 [rat]), and anti-α5 (clone P1D6 [mouse]), all from Merck Millipore; anti-α6 MAbs (clones GoH3 [rat] and MP 4F10 [mouse]) from Abcam; MAb anti-αV (clone 272-17E6 [mouse]) from Thermo Fisher Scientific; MAb anti-β1 (clone P5D2 [mouse]) and PAb anti-β1 (AF1778 [goat]), both from R&D Systems; MAb anti-β3 (clone MHF4 [mouse]) from Abnova; MAb anti-β4 (clone 422325 [mouse]) from R&D Systems; and
Techniques: Infection, Binding Assay, Virus, Immunofluorescence, Labeling, Control, Positive Control

Journal: Journal of Virology
Article Title: Human Adenovirus Type 37 Uses α V β 1 and α 3 β 1 Integrins for Infection of Human Corneal Cells
doi: 10.1128/JVI.02019-16
Figure Lengend Snippet: Effect of penton base-derived, RGD-containing peptides (4 mM) on HAdV-5 and HAdV-37 infection of (A) and binding to (B) HCE cells. 37-RGD, peptide mimicking RGD-containing loop of HAdV-37 penton base. 37-AAA, RGD-containing loop in HAdV-37 penton base peptide replaced with AAA. GRGDSP, peptide used previously to inhibit HAdV infection of human cells. The results are presented as a percentage of the value for the control (i.e., untreated cells). *, P < 0.05; **, P < 0.01; ***, P < 0.001. n = 3.
Article Snippet: The integrin-specific antibodies used for flow cytometry, in situ immunohistochemistry, colocalization, binding, and infection competition analyses were the following: MAbs anti-α2 (clone P1E6 [mouse]), anti-α3 (clones P1B5 [mouse] and ASC-1 [mouse]), anti-α4 (clones P4C2 [mouse] and PS/2 [rat]), and anti-α5 (clone P1D6 [mouse]), all from Merck Millipore; anti-α6 MAbs (clones GoH3 [rat] and MP 4F10 [mouse]) from Abcam; MAb anti-αV (clone 272-17E6 [mouse]) from Thermo Fisher Scientific; MAb anti-β1 (clone P5D2 [mouse]) and PAb anti-β1 (AF1778 [goat]), both from R&D Systems; MAb anti-β3 (clone MHF4 [mouse]) from Abnova; MAb anti-β4 (clone 422325 [mouse]) from R&D Systems; and
Techniques: Derivative Assay, Infection, Binding Assay, Control

Journal: Journal of Virology
Article Title: Human Adenovirus Type 37 Uses α V β 1 and α 3 β 1 Integrins for Infection of Human Corneal Cells
doi: 10.1128/JVI.02019-16
Figure Lengend Snippet: HAdV-37 virions colocalize with α3 and αV integrins on HCE cells. Alexa Fluor 555-labeled HAdV-37 virions (in orange) were incubated with HCE cells at 37°C for different time points and subsequently stained for α3 (P1B5) or αV (272-17E6) integrins (in green). (A) Representative confocal images of HAdV-37 virions colocalizing with integrin antibodies after 15 min (top, α3; bottom, αV). Slices 90° tilted show optical sections of the x-z and y-z planes at the bottom and right side, respectively, of each image on the left. The marked squares display a colocalization event, enlarged to the right in the center of the crosshairs in each box and in the x-y , x-z , and y-z sections. Scale bars, 5 μm. (B) Quantification of the percentage of colocalized HAdV-37 virions with α3 and αV integrins at different time points in HCE cells. Data were collected from 5 to 7 z stacks with at least 2 cells per stack. #, Bars represent data from a pixel shift analysis to test if colocalization occurs by chance. *, P < 0.05. n = 1.
Article Snippet: The integrin-specific antibodies used for flow cytometry, in situ immunohistochemistry, colocalization, binding, and infection competition analyses were the following: MAbs anti-α2 (clone P1E6 [mouse]), anti-α3 (clones P1B5 [mouse] and ASC-1 [mouse]), anti-α4 (clones P4C2 [mouse] and PS/2 [rat]), and anti-α5 (clone P1D6 [mouse]), all from Merck Millipore; anti-α6 MAbs (clones GoH3 [rat] and MP 4F10 [mouse]) from Abcam; MAb anti-αV (clone 272-17E6 [mouse]) from Thermo Fisher Scientific; MAb anti-β1 (clone P5D2 [mouse]) and PAb anti-β1 (AF1778 [goat]), both from R&D Systems; MAb anti-β3 (clone MHF4 [mouse]) from Abnova; MAb anti-β4 (clone 422325 [mouse]) from R&D Systems; and
Techniques: Labeling, Incubation, Staining

Journal: Journal of Virology
Article Title: Human Adenovirus Type 37 Uses α V β 1 and α 3 β 1 Integrins for Infection of Human Corneal Cells
doi: 10.1128/JVI.02019-16
Figure Lengend Snippet: HAdV infection (in green) and expression of integrin subunits α3, αV, and β1 (in red) on HCE cells grown as multilayers at the air-liquid interphase. HAdV-5 and -37 were infected from the apical (upper) side. Antibodies used for staining were P1B5 (α3), 272-17E6 (αV), P5D2 (β1), and B025/AD51 (adenovirus hexon). Nuclear stain is shown in blue. Staining with secondary antibodies only was completely negative. White bar, 50 μm. n = 2.
Article Snippet: The integrin-specific antibodies used for flow cytometry, in situ immunohistochemistry, colocalization, binding, and infection competition analyses were the following: MAbs anti-α2 (clone P1E6 [mouse]), anti-α3 (clones P1B5 [mouse] and ASC-1 [mouse]), anti-α4 (clones P4C2 [mouse] and PS/2 [rat]), and anti-α5 (clone P1D6 [mouse]), all from Merck Millipore; anti-α6 MAbs (clones GoH3 [rat] and MP 4F10 [mouse]) from Abcam; MAb anti-αV (clone 272-17E6 [mouse]) from Thermo Fisher Scientific; MAb anti-β1 (clone P5D2 [mouse]) and PAb anti-β1 (AF1778 [goat]), both from R&D Systems; MAb anti-β3 (clone MHF4 [mouse]) from Abnova; MAb anti-β4 (clone 422325 [mouse]) from R&D Systems; and
Techniques: Infection, Expressing, Staining